 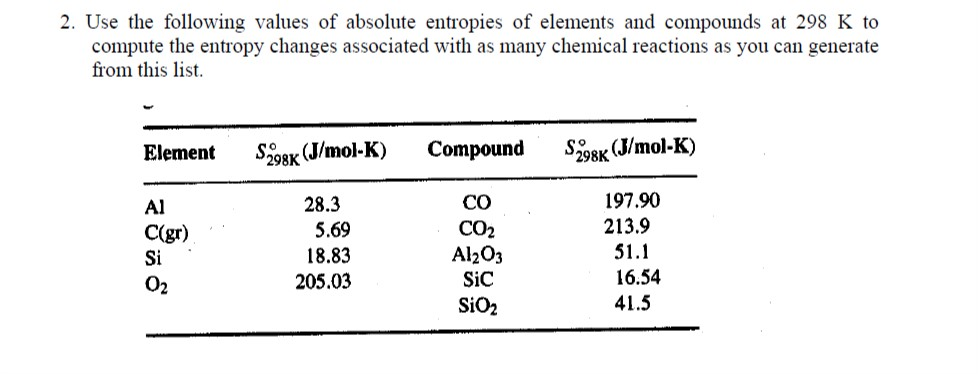
Hence a macroscopic sample of a gas occupies all of the space available to it, simply because this is the most probable arrangement.Įntropy depends not only on the number of atoms or molecules and the volume of available space, but also their freedom of motion, which corresponds to temperature and state of matter. An entropy change of a system is equal to the amount of heat transferred (Qrev) to it in a reversible. The probability of arrangements with essentially equal numbers of molecules in each bulb is quite high, however, because there are many equivalent microstates in which the molecules are distributed equally. Entropy is a measure of molecular disorder or randomness. For processes involving an increase in the number of microstates, Wf > Wi, the entropy of the system increases and S > 0. Although nothing prevents the molecules in the gas sample from occupying only one of the two bulbs, that particular arrangement is so improbable that it is never actually observed. As for other state functions, the change in entropy for a process is the difference between its final ( Sf) and initial ( Si) values: S S f S i k ln W f k ln W i k ln W f W i. Instead of four molecules of gas, what if we had one mole of gas, or 6.022 × 10 23 molecules in the two-bulb apparatus? If we allow the sample of gas to expand spontaneously in the two containers, the probability of finding all 6.022 × 10 23 molecules in one container and none in the other at any given time is extremely small, effectively zero. Take it into your hands, shut your eyes, and twist the sides around randomly a few times.\( \newcommand\): The Possible Microstates for a Sample of Four Gas Molecules in Two Bulbs of Equal Volume Imagine a 2x2 Rubik's cube, solved so that each face contains just one colour.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
